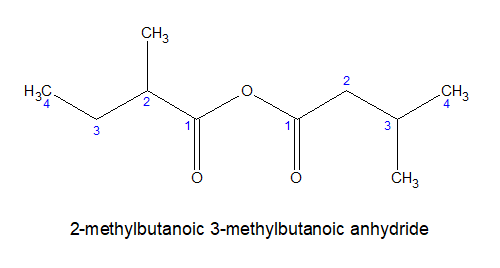
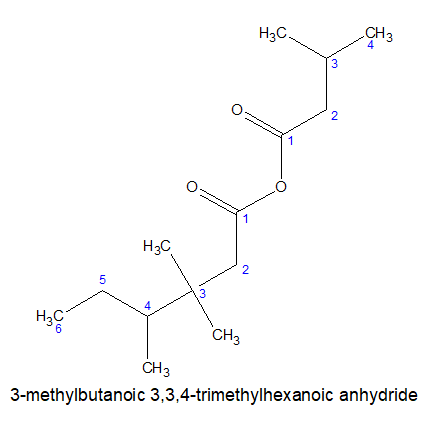
Acid anhydrides are the derivatives of acid formed by removing one water molecule from two identical or different carboxylic acid molecules. If two identical molecules are present it is called simple acid anhydride. If two different molecules are present it is called mixed acid anhydride.
Rules of naming simple acid anhydride:
i) Name the carboxylic acid by normal rule then, instead of writing alkanoic acid, it is written as alkanoic anhydride.

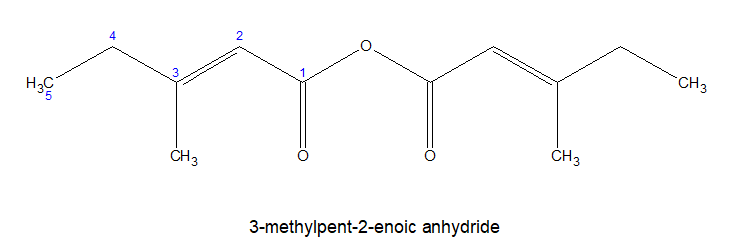
ii)The C of the CO group on either side are considered as 1-locant
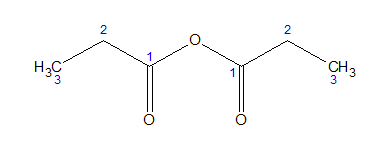

Rules for mixed anhydrides:
i)For mixed anhydrides, the carboxylica acids are named alphabetically