Organic Chemistry is the branch of Chemistry that deals with Carbon and its compound. In this branch we study the preparations and properties of Organic Compounds.
Organic Compounds may be defined as the carbon compounds containing generally hydrogen and one or more few other elements such as oxygen, sulphur, nitrogen, phosphorous, halogens etc.
Unique properties of Carbon:
Carbon atom has some unique property, the other elements do not possess such property. Due to this unique nature of carbon atom, the properties of organic compounds formed from carbon atoms are quite different from the inorganic compounds formed from rest of the elements.
1.Tetravalency:
Valency of carbon is four. It can neither lose or gain electrons rather, it can share electrons.
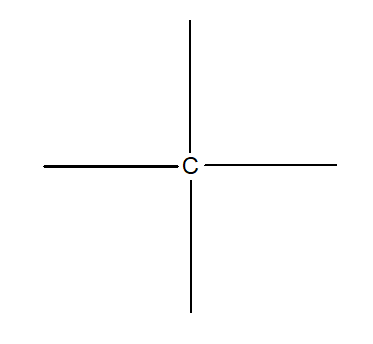
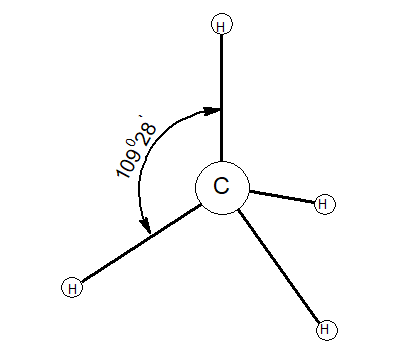
2.Catenation:
Carbon atom has a peculiar property to combine with other carbon atom or atoms by covalent bonds resulting in the formation of a vast number of stable carbon compounds. This unique property of carbon atom to combine with itself is known as Catenation.
The main reasons of this unique property of Carbon are-
- Small size of Carbon atom
- Higher electronegativity of Carbon atom
- High strength of C-C single bonds (Bond Energy 355 kJ/mol) and C-C multiple bonds.

Classification of Organic Compounds
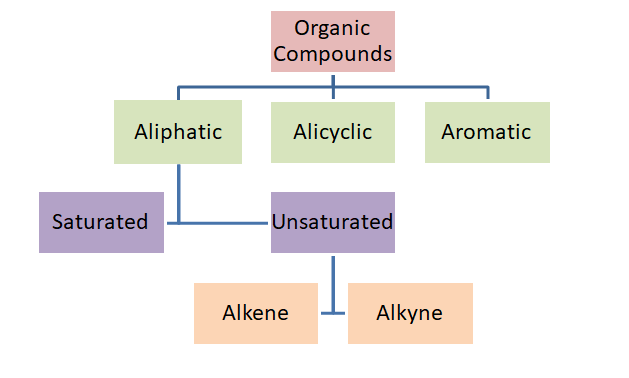
Aliphatic Compounds
Compounds that possess a chain-like structure with or without branching are called aliphatic compounds.
Saturated Compounds
Compounds which possess all single covalent bonds are called saturated compounds. They are also called alkanes.
Unsaturated Compounds
Compounds which possess at least one double or triple bond are called unsaturated compounds. If double bond is present it is called alkene. If only triple bond is present it is called alkyne.
Alicyclic Compounds
Compounds that possess a cyclic ring without alternating single and double bonds are called alicyclic compounds.
Aromatic Compounds
Compounds that possess a cyclic ring with alternating single and double bonds are called aromatic compounds.














